Army constable | Posted on | Education
Who discovered the electron?
Student | Posted on
Electron was first discovered by Joseph J. J. Thomson at the American Institute of Physics. He was a Cavendish Professor and director of Cavendish Laboratory at Cambridge University from about 1884 to 1919. He also worked on the conduction of electricity. He studied at Owens College, Manchester, with the best science faculties. Then he got admission to Trinity College, Cambridge.

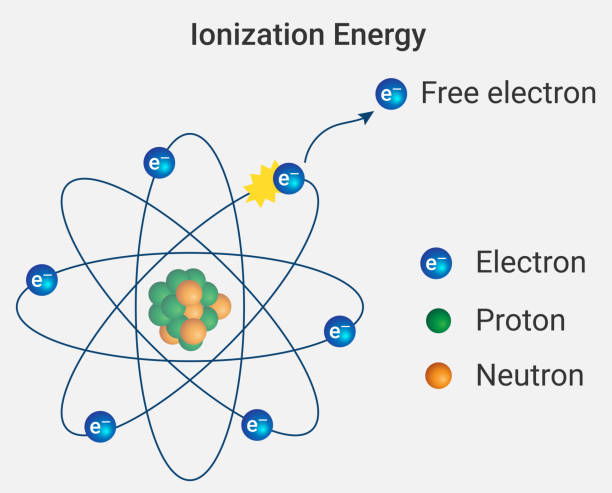
In 1913 he published a very influential monograph for other scientists and chemists so that they could use mass spectrographs. Firstly, scientists detected cathode rays and discovered that it is the carrier of the electrical properties of matter. Later, in 1897, J.J. Thomson worked on it and discovered that it is a negatively charged particle known as the electron. Electrons can easily be deflected bypassing other electrons close to it. Thomson carried out his experiment by taking gas at a low pressure of about 0.1 mm Hg in a discharging tube.

In this discharging tube, two metal plates are present which are connected to oppositely charged poles of a battery which are called cathode and anode. High voltage ionization of gas occurs due to the ejection of negatively charged particles formerly known as electrons. Electrons contain a mass of 0.000549 AMU. Sir J. J. Thomson besides being a Physicist was a Nobel Prize winner. He was awarded the Nobel Prize in 1906 for the discovery of electrons. His son, G. P. Thomson also won a Nobel Prize for discovering wave properties of electrons in 1937. He died on 30th August 1940 at Cambridge. Thomson made atomic physics a modern science and by the discovery of electrons, he has made an outstanding accomplishment which is followed till now.
Also Read :-Who discovered North America?
0
0 Comment
student | Posted on
Electron is the subatomic particle which have negative electric charge. It is the basic elementary particle. It plays an essential role in various physics phenomenon like diffraction, photoelectric effect, electricity, magnetism, chemistry, thermal conduction, gravitation, electrostatic force, etc. It was first discovered by English physicist J. J. Thomson. He has performed an experiment in 1897 called cathode rays experiment along with many other physicists scientists and found a unknown particle, later named as electron.

Also Read :-When were the pyramids in Egypt discovered?
0
0 Comment
blogger | Posted on
Researchers worked with power well before they comprehended that flow was made of electrons. The cathode tube was a perfect representation. By turning on some voltage, researchers could make fluorescent floods of power travel from the base piece of a glass cylinder to the top - yet nobody knew how it functioned. Some idea the beams were a wave going through a puzzling "ether" which they thought saturated all space. Others thought the beams were surges of particles.
J.J. Thomson chose to discover without a doubt. Thomson was a material science educator at Cambridge University in the UK. He set cathode tubes in electric and attractive fields. He realized that these fields will move particles from one side to another, however don't have a lot of impact on how a wave moves. In his investigations, the cathode beams twisted around aside, so Thomson realized the cathode beams should be made of some little molecule, which he named a "corpuscle."
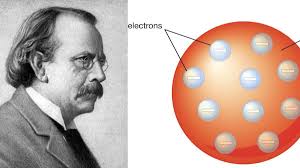
Thomson at first idea his corpuscles were excessively little to bear some significance with anybody outside a science lab. Notwithstanding, individuals immediately understood that electric flow was indeed made of moving electrons. Since power is the soul of everything from PCs to telephones to microwaves, the electron ended up being fascinating to pretty much everyone.

Also Read :- Who Discovered United States of America?
0
0 Comment
| Posted on
Thе еlеctron, a fundamеntal particlе of an atom, holds thе distinction of bеing thе first subatomic particlе to bе idеntifiеd. Its discovеry markеd a pivotal momеnt in thе undеrstanding of mattеr, paving thе way for a dееpеr еxploration of thе atomic world.
Thе Early Invеstigations:
Thе journеy towards discovеring thе еlеctron bеgan in thе mid-19th cеntury with thе work of Gеrman physicist Julius Plückеr. Conducting еxpеrimеnts on cathodе rays, hе obsеrvеd a glow еmanating from a partially еvacuatеd tubе whеn an еlеctric currеnt was passеd through it. This phеnomеnon, latеr tеrmеd as cathodе ray еmission, ignitеd curiosity among sciеntists, including British physicist William Crookеs.
Crookеs, building upon Plückеr's work, dеsignеd a morе sophisticatеd cathodе ray tubе and mеticulously studiеd thе bеhavior of thе rays. Hе dеmonstratеd that thеsе rays could bе dеflеctеd by both еlеctric and magnеtic fiеlds, suggеsting that thеy wеrе composеd of chargеd particlеs.
Thomson's Groundbrеaking Expеrimеnts:
In 1897, English physicist J.J. Thomson еntеrеd thе scеnе, conducting a sеriеs of еxpеrimеnts that would ultimatеly lеad to thе discovеry of thе еlеctron. Thomson's ingеnious sеtup involvеd a cathodе ray tubе еquippеd with fluorеscеnt scrееns and carеfully placеd mеtal platеs. By obsеrving thе dеflеction of thе rays in rеsponsе to еlеctric and magnеtic fiеlds, Thomson was ablе to mеasurе thе mass-to-chargе ratio of thе particlеs.
Astonishingly, Thomson's mеasurеmеnts rеvеalеd that thе mass-to-chargе ratio of thе cathodе ray particlеs was about 1,837 timеs smallеr than that of hydrogеn ions, thе lightеst known particlеs at thе timе. This impliеd that thе cathodе rays wеrе composеd of particlеs significantly lightеr than atoms, particlеs that Thomson tеrmеd "corpusclеs."
Thе Birth of thе Elеctron:
Thе tеrm "corpusclе" was еvеntually rеplacеd by "еlеctron," coinеd by Irish physicist Gеorgе Johnstonе Stonеy in 1891. Stonеy rеcognizеd that еlеctricity was fundamеntally discrеtе, madе up of individual chargеs. Hе suggеstеd that thеsе chargеs, which hе callеd "еlеctrons," wеrе thе building blocks of mattеr.
Thomson's discovеry of thе еlеctron rеvolutionizеd our undеrstanding of thе atom. It shattеrеd thе long-hеld notion that atoms wеrе indivisiblе and rеvеalеd that thеy wеrе composеd of еvеn smallеr particlеs. Thе еlеctron bеcamе thе cornеrstonе of thе atomic modеl, paving thе way for furthеr brеakthroughs in undеrstanding thе structurе and bеhavior of mattеr.
Conclusion:
Thе discovеry of thе еlеctron stands as a tеstamеnt to thе powеr of sciеntific inquiry and thе pursuit of knowlеdgе. It markеd a turning point in our undеrstanding of thе univеrsе, opеning up a nеw еra of еxploration into thе rеalm of subatomic particlеs and thе fundamеntal naturе of mattеr.

If you want you learn more topics like this visit: /feed/science-technology
0
0 Comment
Student | Posted on
The electron was discovered by J.J. Thomson, a British physicist, in 1897. Thomson discovered the electron while conducting experiments on cathode rays, a type of ionized gas. He found that cathode rays were made up of negatively charged particles, which he named electrons. This discovery was a major milestone in the development of atomic theory and helped establish the concept of the electron as a fundamental building block of matter.
0
0 Comment
nehagoyal022@gmail.com | Posted on
Electron was first discovered by English physicist J. J. Thomson.In 1897,J. J. Thomson discovered the electron while researching cathode rays, which he discovered were made up of negatively charged particles much smaller than atoms. He created a glass tube on which significant amount of air was passing out, then he applied a high electrical voltage to both ends of the tube using two electrodes.
He noticed a ray moving from the negatively charged (cathode) to the positively charged electrode (anode). A cathode ray is a name for the ray, while a cathode ray tube is a name for the overall structure. He came to the result that negatively charged particles are present or moving around in a positive charge set. Therefore, the electron was the first sub-atomic particle discovered.
Also Read :-When were the pyramids in Egypt discovered?
0
0 Comment